Categories
The Benefits of Platelet-Rich Fibrin
Key points
- PRF is similar to PRP except that PRF naturally contains fibrin for clot scaffolding and localization of mesenchymal stem cells.
- PRF formation and centrifugation differs from PRP in that there is no anticoagulant and spin times and speeds differ.
- PRF has implied use as a natural filler.
- PRF has had remarked success as a complementary component in surgical and nonsurgical esthetic treatments.
- PRF releases platelet-related therapeutic granules for a longer duration and at a slower rate than PRP.
A new front in medical therapies
Wound healing and tissue regeneration are fundamental goals of medical care. In this context, the use of autologous blood concentrates has emerged. Historically, the primary use of such therapy was in oral maxillofacial surgery1: however, its use in surgical and noninvasive esthetic procedures has shown notable success, suggesting a bright future for esthetic and reconstructive medicine.
Autologous platelet therapy gained popularity in the 1990s with the use of platelet-rich plasma (PRP), which has since found several medical applications. The focus of this article is the next generation of autologous blood concentrate therapy, platelet-rich fibrin (PRF), and its roles in esthetic medicine. The significance of these developments will become apparent throughout the review of the composition of whole blood.
Whole blood composition
Blood is composed of plasma (55%) and cells (45%)2. Plasma consists mostly of water (92%), as well as soluble proteins, electrolytes, and metabolic wastes. The most notable soluble constituent is fibrinogen, a clotting protein. When tissue and vascular injury occur, thrombin enzymatically converts fibrinogen to insoluble fibrin.2, 3, 4, 5 Fibrin then acts as the binding scaffold for platelets and erythrocytes in clot formation, which is the essential first step in wound healing and tissue regeneration.1, 2, 3, 4 Beyond plasma, red blood cells (erythrocytes), white blood cells (leukocytes), and platelets (thrombocytes) constitute the remaining cellular component of whole blood2. Erythrocytes are the most abundant, comprising about 44% of total blood composition, whereas leukocytes and thrombocytes constitute the buffy coat at less than 1%.2
Centrifuging whole blood conveniently separates its components according to density. Erythrocytes collect at the bottom of the tube, forming the hematocrit layer; the thin, white-tinted buffy coat settles at the top of the erythrocytes; and plasma forms the supernatant.1, 2, 6 Varying centrifugation speed and duration further separates blood concentrate components. Anticoagulants or enzymatic supplements may be required to separate PRP and platelet-poor plasma (PPP),1, 7 the former with enough platelets for therapeutic use, yet less abundant than PPP at roughly 25% and 75% of the supernatant volume, respectively.8
What is autologous blood concentrate therapy?
First, There Was PRP
The widely accepted mechanism of PRP therapy is growth factor secretion from platelet alpha granules. When activated in vivo through injury and clot formation, alpha factors bind to the platelet surface and release platelet-derived growth factors, transforming growth factors, fibroblastic growth factor, epithelial cell growth factor, insulin-like growth factor, and vascular endothelial growth factor (Table 1).1, 8, 9, 10 Collectively, these signals help stimulate mesenchymal stem cell (MSC) migration and differentiation at the site of clot formation.1, 11, 12 For PRP, induced clot formation localizes growth factor secretion to its implemented site.1, 13
Table 1. Platelet therapy growth factor functions
Platelet-derived growth factor (PDGFaa, PDGFbb, PDGFab)
- Triggers the activities of neutrophils, fibroblasts, and macrophages
- Chemoattractant/cell proliferator
- Stimulates mesenchymal cell lineages
Transforming growth factor (TGFβ1, TGFβ2, TGFβ3)
- Promotes cellular differentiation and replication
- Stimulates matrix and collagen synthesis
- Stimulates fibroblast activity and collagen production
Vascular endothelial growth factor (VEGF)
- Angiogenesis
- Stimulates synthesis of basal lamina
Fibroblastic growth factor (FGF)
- Angiogenesis
- Fibroblast production
Epithelial cell growth factor (ECGF)
- Stimulates epithelial cell replication
Insulin-like growth factor (IGF-1)
- Promotes cellular growth and proliferation
In aging skin, PRP’s targeted growth factor secretion promotes fibroblast proliferation and gene expression that stimulates type I collagenesis14. To harness these abilities, PRP must first be produced using anticoagulant.6, 10 Second, to ensure that platelet activation and fibrin clot formation occurs, calcium chloride and thrombin, which is often bovine derived, must be added to the PRP preparation6, 9, 10, 13, 15; using bovine-derived thrombin poses the risk of inducing adverse immunologic reactions.6, 16 Alternatively, additives can be omitted and trust that activation and clot formation occur spontaneously in vivo13, which is not guaranteed. If PRP’s production process potentially compromises its benefits, then its antiaging properties may also be at risk. Fortunately, PRF is a readily available promising alternative.
The Next Generation of Autologous Platelet Therapy: Platelet Rich Fibrin
PRF was first introduced in 2000 by Joseph Choukroun and colleagues17. PRF offers all the clinical benefits of PRP as well as a naturally forming fibrin scaffold that guides clot formation, serves as a supportive template for tissue regeneration, and that sustains growth factors and stem cells.6, 10, 18.
In contrast to PRP, PRF is obtained by centrifuging whole blood without any additives.10, 15 Without anticoagulant, PRF spontaneously forms a fibrin matrix gelatinous clot9, 10, 15 that confines growth factor secretion to the clotting site. In tissue repair, recruited fibroblasts reorganize this fibrin matrix and initiate collagen synthesis19. Thus, the combined effects of growth factor secretion and fibroblast recruitment in PRF work synergistically to promote collagenesis and tissue regeneration.
Injury-induced growth factor signaling recruits MSCs to the compromised site11, 12, 20, 21 where they subsequently differentiate.11, 18, 21 Surgeries and injections simulate local injury and trigger the same signaling cascade. Applying PRF with these treatments localizes and enhances the regenerative processes spurred by the body’s natural response to injury. In the context of attracting, entrapping, and sustaining MSCs, research has also revealed that fibrin serves as a successful culture medium and carrier of MSCs,18, 22, 23 which preserves the paracrine functions essential in conferring their regenerative effects24.
PRF also sustains other vital cells, including leukocytes.18 Analyzing the cellular content of the PRF clot reveals that most leukocytes within a whole-blood sample are contained within PRF after centrifugation.10, 25 These leukocytes secrete signaling factors that further stimulate tissue repair1, 26 and MSC recruitment.12
MSCs bear important regenerative applications; their multipotency allows them to give rise to several tissues including bone, cartilage, adipose, dermis, and other mesodermal tissues.27, 28, 29 MSCs in peripheral blood have been isolated30, 31 and shown to proliferate and differentiate on stimulation.21, 32 Di Liddo and colleagues33 detected in vitro multipotent stem cell markers within PRF, and reported that a fraction of cells in PRF express defining phenotypic features of MSCs. Therefore, PRF establishes a local environment conducive to MSC migration and may also serve as a stem cell source.
Altering centrifugation duration and speed allows for manipulation of product volume and clotting onset. Broadly, this yields 2 categories of PRF:
1. Injectable PRF: clot formation occurs around 15 minutes postcentrifugation34
2. PRF that coagulates during centrifugation: this is most useful when a biological membrane or a physiologic glue is needed (Fig. 1)
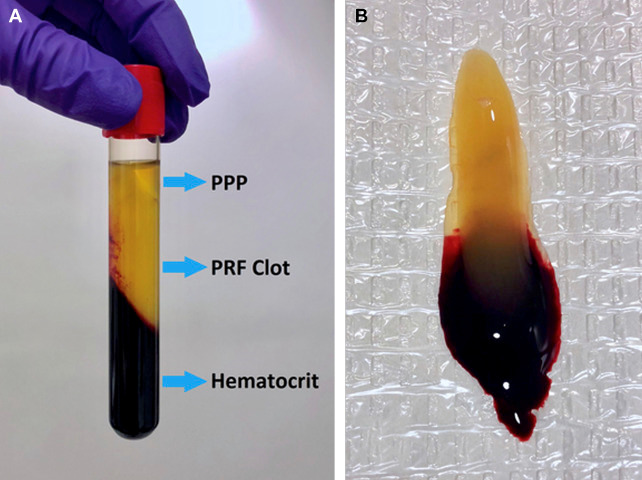
Fig. 1. (A) Visualization of the resulting blood concentrate layers from centrifugation to immediately procure a PRF clot.
(B) The PRF clot removed from the centrifuge tube.
Platelet-Rich Plasma Versus Platelet Rich Fibrin
PRF has various advantages over PRP. With PRP and other past-generation platelet concentrates, growth factor release is initially rapid, yielding short-lived, early healing benefits without long-term improvement. The relatively short half-lives of growth factors,13, 35 in conjunction with their abundant and rapid release following PRP activation36, supports this lack of prolonged efficacy, because tissue receptor saturation may prevent additional growth factors from binding a receptor before their degradation. Recall that preparing functional PRP requires external additives, which brings uncertainty about its spontaneous activation in vivo. Thus, functional PRP is either nonautologous, or its efficacy is unguaranteed.6
Contrastingly, PRF requires no additives. Activation and fibrin clot formation are based on known, intrinsic properties of blood, and the timeliness of activation is relatively well understood. Furthermore, the holistically autologous nature of PRF reduces the risk of immunogenic reaction and disease transmission.6
Most notably, in comparison with PRP’s rapid growth factor release, PRF releases growth factors15 for an extended duration of time: up to 7 days for most growth factors, and even longer for others. It is proposed that PRF’s composition aids in preventing rapid proteolysis of growth factors, thereby enabling prolonged secretion. In addition, the slow polymerization and remodeling of the fibrin matrix within PRF, compared with PRP’s more rapid, haphazard polymerization, effectively sustains growth factors and other critical cells.7, 9, 15 Masuki and colleagues concluded from their comparative analysis that growth factor concentrations are generally higher in PRF than PRP, a finding that supports PRF’s marked efficacy in stimulating angiogenesis, wound healing, and tissue regeneration. As previously mentioned, growth factors chemotactically attract MSCs. Therefore, it is reasonably assumed that PRF’s sustained growth factor secretion, in comparison with PRP, more strongly induces MSC migration to the site of its application, a conclusion that is further backed by comparative in vitro studies.37,40
Beyond their chemotactic and compositional differences, PRF and PRP undergo different centrifugation premises for their procurement (Fig. 2). The low-speed centrifugation of PRF tends to better preserve the beneficial cellular content within the resulting PRF layer,26, 34, 41 whereas high-speed centrifugation, such as that seen in the hardspin stage of PRP preparation, tends to push most cells to the bottom of the tube 26

Fig. 2. Separations achieved by each blood concentrates’ respective centrifugation parameters. The white separating gel and anticoagulant necessary for preparing PRP is visualized at the bottom of the whole blood tube and is pictured separating PRP and the hematocrit in the PRP postcentrifugation tube. (Courtesy of CosmoFrance, Inc., Miami, FL.)
Regarding cost, PRP incurs the cost of separating gel, anticoagulant, and activation additives, whereas PRF does not. PRF use implies reduced costs for provider and patient alike.
Applications of platelet rich fibrin
The following text outlines a few of the many applications of PRF in cosmetic medicine and surgery. Other applications not discussed, but worth mentioning, include enhanced healing following ablative skin resurfacing laser treatments and collagen induction with microneedling.
Natural Filler
Aging skin naturally loses collagen, elasticity, and volume. The dermis thins and the fibroblast population declines, reducing the production of collagen and hyaluronic acid. As collagen decreases by approximately 1% yearly, skin laxity and wrinkling become apparent.14, 43 Our skin also loses moisture because the hydrophilic hyaluronic acid concentration declines. Consequently, the dermis loses turgidity, resulting in volume loss and unesthetic changes. Stimulating collagenesis and hyaluronic acid content within aging skin may overcome these changes. Here, PRF shows promise.
With high concentrations and slow release of fibroblastic growth factors, PRF, when injected beneath the skin, should stimulate fibroblast formation and subsequently increase collagen and hyaluronic acid content. PRF also enmeshes hyaluronic acid, sustaining it where injected. As it forms a gel, PRF produces an immediate volumization effect; although this volumization lasts only a few weeks, repeated treatments yield long-term effects from prolonged collagen production and localized regenerative activity.
In his own practice, the primary author has found success with injecting PRF alone as a natural, autologous dermal filler, which has resulted in volume restoration of hollowing tear troughs, improved fine lines, and homogenization of pigmentation irregularities with repeat treatments (Fig. 3).
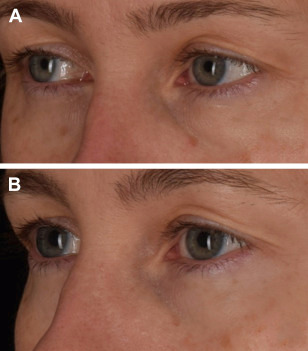
Fig. 3. A 45-year-old female patient (A) before and (B) after 3 treatments of infraorbital PRF injections spaced 4 to 6 weeks apart to correct pigmentation irregularities, stimulate volume restoration, improve fine lines, and reduce under-eye hollowing
The author has also treated patients with a combination of PRF and hyaluronic acid filler (Fig. 4, Video 1). Together, PRF and filler synergistically improve moisture retention and create a scaffold for collagen growth as the body metabolizes the filler over time. The author has anecdotally found that a concentration of 2 parts filler to 1 part PRF will lead to a sustained filler effect and still harness the advantages of the PRF.
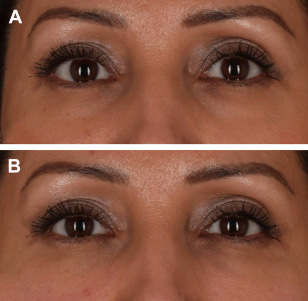
Fig. 4. (A) Before and (B) immediately after treatment of a 40-year-old female patient with hyaluronic acid filler and PRF injected to infraorbital hollows.
Fat Grafting
Autologous fat transfer, although slightly more invasive than office-based hyaluronic acid fillers, efficaciously restores volume loss. Unlike conventional dermal fillers, fat grafts provide potentially permanent volume restoration; however, only roughly half of the transferred cells survive. Fortunately, PRF shows promise in improving fat retention (Fig. 5).
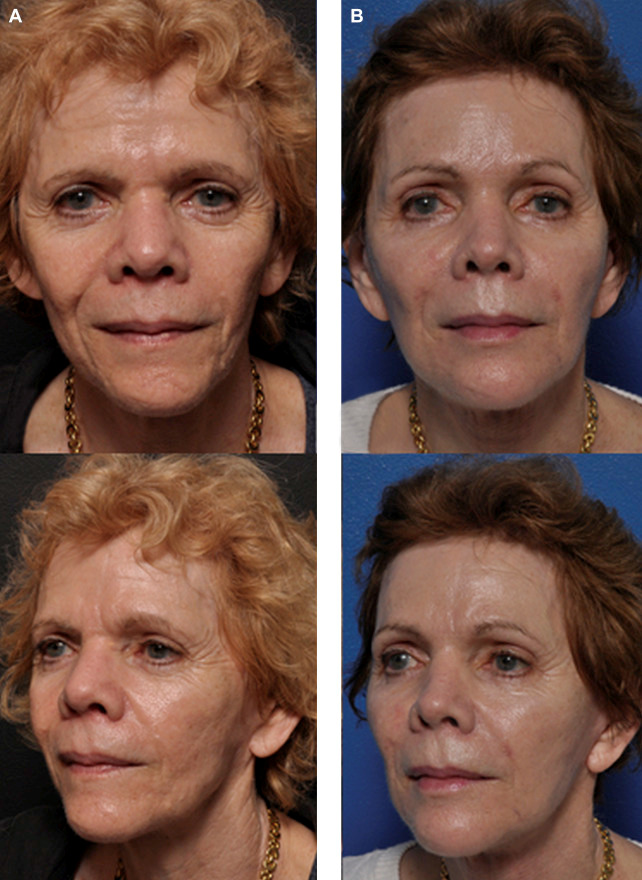
Fig. 5. (A) Before and (B) 3 months after fat transfer supplemented with PRF, an endoscopic brow lift, and a face lift were performed on this 66-year-old female patient.
Adipose tissue is considered an exceptional stem cell source. Furthermore, subcutaneous fat is an especially attractive source of progenitor cells because of its accessibility, abundance, and the existence of a supportive stromal vascular fraction (SVF). The SVF of the abdominal subcutaneous tissues is regarded as an exceptional fat harvesting location considering its abundant supply of adipose-derived stem cells. However, stem cell viability can be difficult to sufficiently sustain while in transit during fat transfer. Liu and colleagues highlighted the enhancement of fat transfer with PRF supplementation, detailing that implementing PRF reduced resorption and improved retention of fat grafts. PRF’s effect on fat survival likely results from its prolonged growth factor release and the ability of the autologous fibrin matrix to sufficiently support stem cell transfer. A study by Keyhan and colleagues supported this premise, reporting that PRF more effectively improved fat graft retention than PRP. Video 2 depicts the process conducted to supplement fat transfer with PRF
Facial Surgery
As invasive treatments, facial surgeries elicit strong clotting and wound-healing responses. The resulting blood clots consist mainly of erythrocytes. Applying PRF to the surgical site effectively replaces the abundance of erythrocytes with fibrin, leukocytes, stem cells, and platelet-derived growth factors. This results in accelerated wound healing and attraction of MSCs to the site, laying the foundation for tissue regeneration, collagen remodeling, and a sustained cosmetic result.
In rhinoplasties, cartilage grafts are frequently needed to achieve optimal results. Cartilage grafts are formed from diced autologous or cadaver cartilage. When used alone, diced cartilage may scatter after placement, resulting in palpable or visible structural irregularities.50, 51 PRF aids in forming and depositing cartilage grafts by acting as a physiologic glue that enhances the consistency and pliability of the grafts and reduces the probability of graft rejection owing to its autologous nature (Video 3). Studied in a rabbit model, PRF effectively improved cartilage graft viability, and, in another study, it stimulated cartilage regeneration more so than PRP
Hair Loss and Scar Therapy
The primary author has found that PRF injections improve scar appearance and stimulate hair growth where hair follicles are present but inactive (Fig. 6).
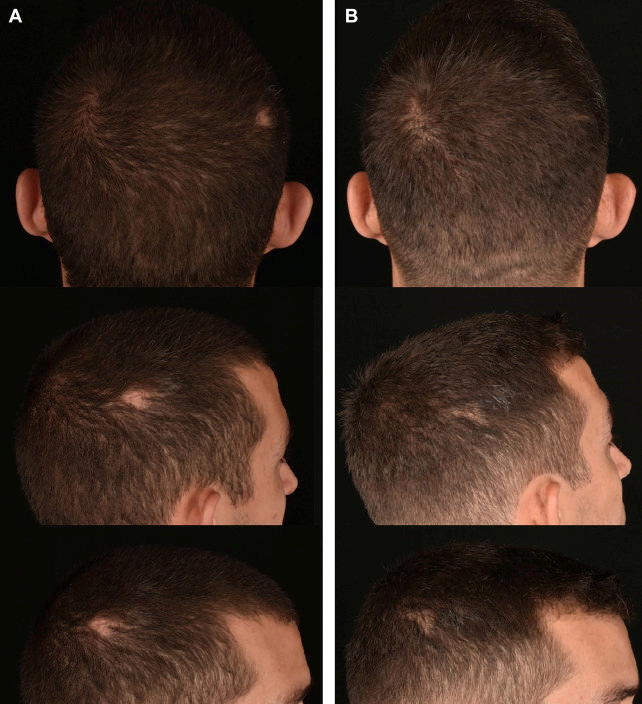
Fig. 6. (A) Before and (B) after 1 session of PRF injections to treat the appearance and hair growth of a lateral parietal trauma-induced scar of a 28-year-old male patient. Progress photos were obtained 6 months after treatment. Before treatment, hair growth was dormant for 5 weeks
The future of platelet rich fibrin
The widespread applications of PRF solidify its place among autologous blood concentrate therapies as both a primary and supplementary medical tool. Further research is expected to uncover additional benefits to be obtained from PRF’s bioavailability, autologous nature, and regenerative properties.
Supplementary data
References
1. A.K. Garg
Autologous blood concentrates
Quintessence Publishing Co. Inc., Batavia (IL) (2018)
Google Scholar
2. A.L. Mescher
Blood
Junqueira's basic histology text & atlas (15th edition), McGraw-Hill, New York (2013)
Chapter 12
Google Scholar
3. K.E. Brummel, S. Butenas, K.G. Mann
An integrated study of fibrinogen during blood coagulation
J Biol Chem, 274 (32) (1999), pp. 22862-22870
4. K.G. Mann, K. Brummel, S. Butenas
What is all that thrombin for?
J Thromb Haemost, 1 (7) (2003), pp. 1504-1514
5. A. Undas, R.A. Ariëns
Fibrin clot structure and function
Arterioscler Thromb Vasc Biol, 31 (12) (2011), pp. 88-99
6. D.N. Utomo, F. Mahyudin, K.D. Hernugrahanto, et al.
Implantation of platelet rich fibrin and allogenic mesenchymal stem cells facilitate the healing of muscle injury: an experimental study on animal
Int J Surg Open, 11 (2018), pp. 4-9
7. D.M. Dohan, J. Choukroun, A. Diss, et al.
Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologi features
Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 101 (3) (2006), pp. 45-50
8. R.E. Marx, E.R. Carlson, R.M. Eichstaedt, et al.
Platelet-rich plasma: growth factor enhancement for bone grafts
Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 85 (6) (1998), pp. 638-646
9. D.M. Dohan, J. Choukroun, A. Diss, et al.
Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution
Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 101 (3) (2006), pp. 37-44
10. D.M.D. Ehrenfest, L. Rasmusson, T. Albrektsson
Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and plateletrich fibrin (L-PRF)
Trends Biotechnol, 27 (3) (2009), pp. 158-167
11. H.D. Boer, C. Verseyden, L. Ulfman, et al.
Fibrin and activated platelets cooperatively guide stem cells to a vascular injury and promote differentiation towards an endothelial cell phenotype
Arterioscler Thromb Vasc Biol, 26 (7) (2006), pp. 1653-1659
12. A.L. Ponte, E. Marais, N. Gallay, et al.
The in vitro migration capacity of human bone marrow mesenchymal stem cells: comparison of chemokine and growth factor chemotactic activities
Stem Cells, 25 (7) (2007), pp. 1737-1745
13. C. Cavallo, A. Roffi, B. Grigolo, et al.
Platelet-rich plasma: the choice of activation method affects the release of bioactive molecules
Biomed Res Int, 2016 (2016), pp. 1-7
14. D.H. Kim, Y.J. Je, C.D. Kim, et al.
Can platelet-rich plasma be used for skin rejuvenation? Evaluation of effects of platelet-rich plasma on human dermal fibroblast
Ann Dermatol, 23 (4) (2011), pp. 424-431
15. D.M.D. Ehrenfest, G.M.D. Peppo, P. Doglioli, et al
Slow release of growth factors and thrombospondin-1 in Choukrouns platelet-rich fibrin (PRF): a gold standard to achieve for all surgical platelet concentrates technologies
Growth Factors, 27 (1) (2009), pp. 63-69
16. V.S. Raja, E.M. Naidu
Platelet-rich fibrin: evolution of a second-generation platelet concentrate
Indian J Dent Res, 19 (1) (2008), pp. 42-46
17. J. Choukroun, F. Adda, C. Schoeffer, et al.
PRF: an opportunity in perio-implantology
Implantodontie, 42 (2000), pp. 55-62
18. J. Choukroun, A. Diss, A. Simonpieri, et al.
Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing
Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 101 (3) (2006), pp. 56-60
19. T.-L. Tuan, A. Song, S. Chang, et al.
In VitroFibroplasia: matrix contraction, cell growth, and collagen production of fibroblasts cultured in fibrin gels
Exp Cell Res, 223 (1) (1996), pp. 127-134
20. E.V. Badiavas, M. Abedi, J. Butmarc, et al.
Participation of bone marrow derived cells in cutaneous wound healing
J Cell Physiol, 196 (2) (2003), pp. 245-250
21. L. Xu, G. Li
Circulating mesenchymal stem cells and their clinical implications
J Orthop Translat, 2 (1) (2014), pp. 1-7
22. W. Bensaïd, J. Triffitt, C. Blanchat, et al.
A biodegradable fibrin scaffold for mesenchymal stem cell transplantation
Biomaterials, 24 (14) (2003), pp. 2497-2502
23. I. Catelas, N. Sese, B.M. Wu, et al.
Human mesenchymal stem cell proliferation and osteogenic differentiation in fibrin gels in vitro
Tissue Eng, 12 (8) (2006), pp. 2385-2396
24. I. Kim, S.K. Lee, J.I. Yoon, et al.
Fibrin glue improves the therapeutic effect of MSCs by sustaining survival and paracrine function
Tissue Eng Part A, 19 (21–22) (2013), pp. 2373-2381
25. D.M.D. Ehrenfest, M.D. Corso, A. Diss, et al.
Three-dimensional architecture and cell composition of a Choukrouns platelet-rich fibrin clot and membrane
J Periodontol, 81 (4) (2010), pp. 546-555
26. S. Ghanaati, P. Booms, A. Orlowska, et al
Advanced platelet-rich fibrin: a new concept for cell-based tissue engineering by means of inflammatory cells
J Oral Implantol, 40 (6) (2014), pp. 679-689
27. N. Kim, S.-G. Cho
Clinical applications of mesenchymal stem cells
Korean J Intern Med, 28 (4) (2013), pp. 387-402
28. J. Kobolak, A. Dinnyes, A. Memic, et al.
Mesenchymal stem cells: identification, phenotypic characterization, biological properties and potential for regenerative medicine through biomaterial micro-engineering of their niche
Methods, 99 (2016), pp. 62-68 Article Download PDF View
29. A.I. Caplan
Mesenchymal stem cells
J Orthop Res, 9 (5) (1991), pp. 641-650
30. G.Y. Rochefort, B. Delorme, A. Lopez, et al.
Multipotential mesenchymal stem cells are mobilized into peripheral blood by hypoxia
Stem Cells, 24 (10) (2006), pp. 2202-2208
31. T. Tondreau, N. Meuleman, A. Delforge, et al.
Mesenchymal stem cells derived from CD133-positive cells in mobilized peripheral blood and cord blood: proliferation, Oct4 expression, and plasticity
Stem Cells, 23 (2005), pp. 1105-1112
32. P.-P. Chong, L. Selvaratnam, A.A. Abbas, et al.
Human peripheral blood derived mesenchymal stem cells demonstrate similar characteristics and chondrogenic differentiation potential to bone marrow derived mesenchymal stem cells
J Orthop Res, 30 (4) (2011), pp. 634-642
33. R. Di Liddo, T. Bertalot, A. Borean, et al.
Leucocyte and platelet-rich fibrin: a carrier of autologous multipotent cells for regenerative medicine
J Cell Mol Med, 22 (3) (2018), pp. 1840-1854
34. M.A.E. Raouf, X. Wang, S. Miusi, et al.
Injectable-platelet rich fibrin using the low speed centrifugation concept improves cartilage regeneration when compared to platelet-rich plasma
Platelets (2017), pp. 1-9,
35. L. Creaney, B. Hamilton
Growth factor delivery methods in the management of sports injuries: the state of play
Br J Sports Med, 42 (2008), pp. 314-320
36. E. Kobayashi, L. Flückiger, M. Fujioka-Kobayashi, et al.
Comparative release of growth factors from PRP, PRF, and advanced-PRF
Clin Oral Investig, 20 (9) (2016), pp. 2353-2360
37. L. He, Y. Lin, X. Hu, et al
A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro
Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 108 (2009), pp. 707-713
38. R. Lundquist, M.H. Dziegiel, M.S. Ågren
Bioactivity and stability of endogenous fibrogenic factors in platelet-rich fibrin
Wound Repair Regen, 16 (3) (2008), pp. 356-363
39. H. Masuki, T. Okudera, T. Watanebe, et al.
Growth factor and pro-inflammatory cytokine contents in platelet-rich plasma (PRP), plasma rich in growth factors (PRGF), advanced platelet-rich fibrin (A-PRF), and concentrated growth factors (CGF)
Int J Implant Dent, 2 (1) (2016), pp. 1-6
40. M.O. Schär, J. Diaz-Romero, S. Kohl, et al..
Platelet-rich concentrates differentially release growth factors and induce cell migration in vitro
Clin Orthop Relat Res, 473 (5) (2015), pp. 1635-1643
41. M. Fujioka-Kobayashi, R.J. Miron, M. Hernandez, et al.
Optimized platelet-rich fibrin with the low-speed concept: growth factor release, biocompatibility, and cellular response
J Periodontol, 88 (1) (2017), pp. 112-121
42. M.A. Farage, K.W. Miller, P. Elsner, et al.
Characteristics of the aging skin
Adv Wound Care, 2 (1) (2013), pp. 5-10
43. R. Ganceviciene, A.I. Liakou, A. Theodoridis, et al.
Skin anti-aging strategies
Dermatoendocrinol, 4 (3) (2012), pp. 308-319
44. E. Papakonstantinou, M. Roth, G. Karakiulakis
Hyaluronic acid: a key molecule in skin aging
Dermatoendocrinol, 4 (3) (2012), pp. 253-258
45. I. Ghersetich, T. Lotti, G. Campanile, et al.
Hyaluronic acid in cutaneous intrinsic aging
Int J Dermatol, 33 (2) (1994), pp. 119-122
46. L.A. Peer
Loss of weight and volume in human fat grafts
Plast Reconstr Surg, 5 (3) (1950), pp. 217-230
47. W.J.F.M. Jurgens, M.J. Oedayrajsingh-Varma, M.N. Helder, et al
Effect of tissue-harvesting site on yield of stem cells derived from adipose tissue: implications for cellbased therapies
Cell Tissue Res, 332 (3) (2008), pp. 415-426
48. B. Liu, X.-Y. Tan, Y.-P. Liu, et al.
The adjuvant use of stromal vascular fraction and platelet-rich fibrin for autologous adipose tissue transplantation
Tissue Eng Part C Methods, 19 (1) (2013), pp. 1-14
49. S.O. Keyhan, S. Hemmat, A.A. Badri, et al.
Use of platelet-rich fibrin and platelet-rich plasma in combination with fat graft: which is more effective during facial lipostructure?
J Oral Maxillofac Surg, 71 (3) (2013), pp. 610-621
50. A. Göral, C. Aslan, B.B. Küçükzeybek, et al
Platelet-rich fibrin improves the viability of diced cartilage grafts in a rabbit model
Aesthet Surg J, 36 (4) (2016), pp. 153-162
51. R.K. Daniel, J.W. Calvert
Diced cartilage grafts in rhinoplasty surgery
Plast Reconstr Surg, 113 (7) (2004), pp. 2156-2171



